The resulting energy curves, along with the corresponding exact results, are plotted in the left graphic, for either the singlet or triplet states. (We have added an additional effective nuclear charge parameter for greater flexibility.) We have not considered a more systematic procedure, in which the parameters can be determined variationally for each value of. Select the parameters, and "by hand," using the sliders, so as to produce the best visible fit to the energy functions obtained by accurate quantum-mechanical computations. The energy of the hydrogen molecule, referred to the energy of two separated hydrogen atoms ( ), can be written in atomic units as: Le modèle de Bohr de lhydrogène repose sur lhypothèse non-classique que les électrons tournent autour du noyau selon des couches ou orbites spécifiques. For the repulsive triplet state, the orbits are outside the internuclear axis and move in parallel. For the singlet state, the electron orbits are located between the nuclei and their electrons orbit in a contra-rotating sense. The two electrons, shown in red, traverse circular orbits of radii perpendicular to the internuclear axis and centered at distances from their respective nuclei. The two protons, shown in blue on the left graphic, are separated by an internuclear distance. In 1915 he postulated a number of general assumptions:. (They are the same movies as on your CD, animations 8.In this Demonstration, we propose a modified version of Bohr's model for the lowest singlet and triplet states of the hydrogen molecule. Bohr pictured the electron in hydrogen orbiting the central atomic nucleus. Please look on your CD for animations regarding absorbsion and emission or go Light photons emitted are listed on the right side, with the arrow showing the The energy levels (n) are listed on the left side, followed by Bohr model of hydrogen atom postulates An atom or hydrogen atom possesses several stable circular orbitals in which an electron can stay. Shows the various energies and the corresponding transitions.Īn energy level diagram for a hydrogen atom, notĭrawn to scale. The red line comes an electron going from 3 to 2. Moving from a higher energy orbit to a lower energy orbit, it emits a photon ofĪ specific frequency and energy value. Each time an electron males a "quantum leap," While you are there, notice that each of the elements has a different patternĬan use this to analyze what some matter is made of? YES! Red absorption line comes from an electron absorbing that wavelength and going When a broad visible spectrum of light shines on hydrogen, notice it only Look at the following applet and start with hydrogen. Is because the electrons can only be in these distinct orbitals. This explains why hydrogen absorbs only specific wavelengths of light and Transitions to 2 (3 to 2, 4 to 2, 5 to 2 etc) are in the visible region. The transitions to 1 (2 to 1, 3 to 1, 4 to 1 etc) are all in the UV region This experiment it is excited into a higher orbit by the electrical energy (sayīack to a more stable orbit it can release the energy as light of a specific 
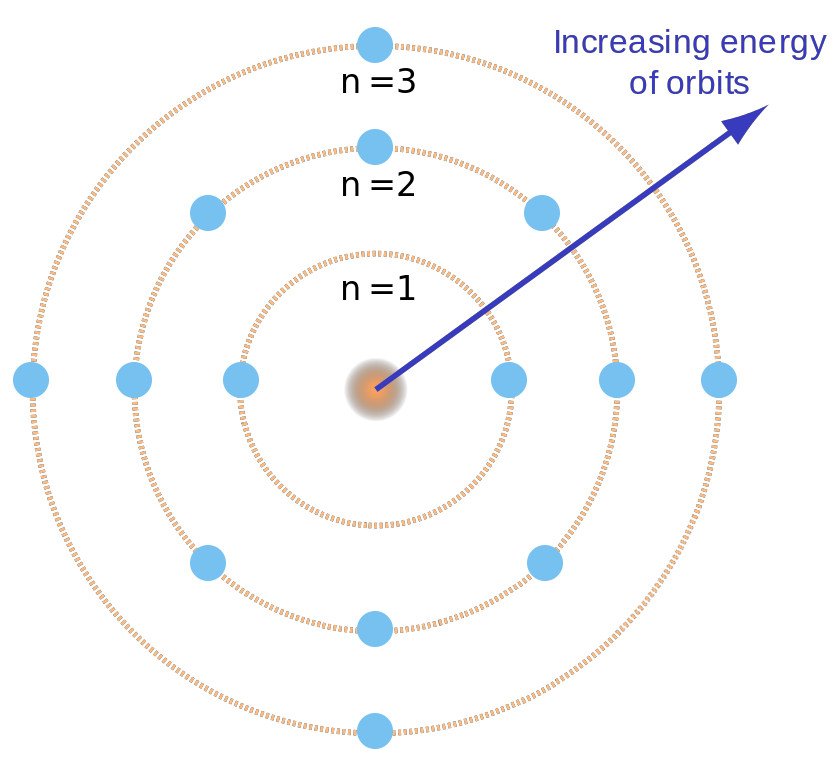
The electron is normally in the lowest orbit (orbit number 1) which That the energies of the electrons are quantized. By saying that the electron can only have specific energies, we can say 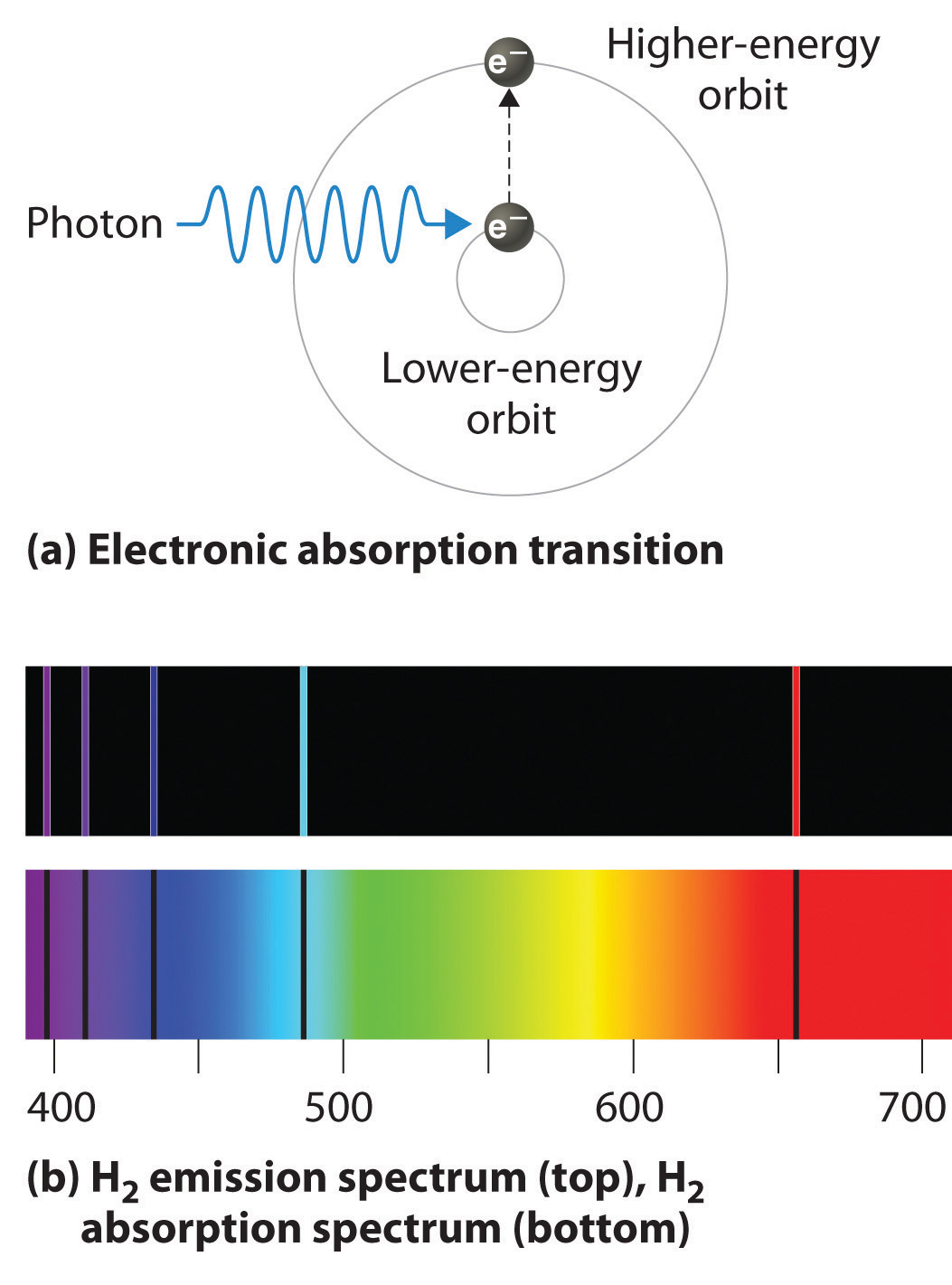
The energy of the orbits increases as you get further from the nucleus. The energy of the electron in an orbit is proportional to its distance from the nucleus. The electron in a hydrogen atom travels around the nucleus in a circular orbit. The Bohr model was based on the following assumptions. What Bohr said was that the electron could only exist in these blue orbits,ĭefinite energy with the inner orbit being the lowest in energy. The Bohr Model of the Atom Niels Bohr proposed a model for the hydrogen atom that explained the spectrum of the hydrogen atom. I have drawn 4 orbits but there are more. An electron in an atom can only exist in certain orbits around the Stated that the electrons orbit the nucleus the way the earth orbits the sun. Wall socket and convert that to radiant or light energy. The lamps get there energy from the electrical potential energy of the Produces a line spectrum that contains only certain frequencies. This does not surprise us as we remember that white light is aĪn interesting thing happens when we use light just from one element, such as Into a spectrum from red (lowest energy) to violet (highest energy). Through a thin slit and then through a prism, the prism will separate the colors Atomic Spectra and the Bohr Model Atomic Spectra and the Bohr Modelįrom incandescent solids, liquids, or dense gases, produces a continuous 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
